Summary
The absorption kinetics of different pharmaceutical formulations of orally and rectally administered flecainide have been assessed in a cross-over study in 7 healthy volunteers. The subjects received single doses of flecainide after a washout period of at least one week. A tablet, an oral solution, a rectal solution and a 10 min i.v. infusion during 10 min each containing 100 mg flecainide were administered to the subjects in a randomized order.
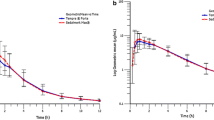
The mean absolute bioavailability was 98%, 78% and 81% for the rectal and oral solutions and the tablet. The lag time after administration of the oral solution was 0.33 h and it was 0.86 h after the tablet and 0.18 h after the rectal solution. The mean time to the peak serum concentration (tmax) after the rectal solution (0.67 h) was shorter than after either the tablet (4 h) or oral solution (1 h). The maximum serum concentration (Cmax) was 0.29 mg · 1−1 after the rectal solution, 0.14 mg · 1−1 after the tablet and 0.17 mg · 1−1 after the oral solution. All the volunteers showed significantly higher serum flecainide concentrations during the first 20 min of the absorption phase after rectal administration of 100 mg flecainide as a solution compared to its oral administration.
In conclusion: based on the absolute bioavailability, Cmax, tmax, and lag times, rectal administration of flecainide solution gave a better absorption profile than after oral tablet or solution.
Article PDF
Similar content being viewed by others
References
Holmes B, Heel RC (1985) Flecainide. A preliminary review of its pharmacodynamic properties and therapeutic efficacy. Drugs 29: 1–33
Camm AJ, Hellestrand KJ, Nathan AW, Bexton RS (1985) Clinical usefulness of flecainide acetate in the treatment of paroxysmal supraventricular arrhythmias. Drugs 29 [Suppl 4]: 7–13
Suttorp MJ, Kingma JH, Lie-A-Huen L, Mast EG (1989) Intravenous flecainide versus verapamil for acute conversion of atrial fibrillation or atrial flutter to sinus rhythm. Am J Cardiol 63: 693–696
Lie-A-Huen L, Kingma JH (1988) Rectal absorption of flecainide acetate. Eur J Clin Pharmacol 35: 89–91
Conard GJ, Carlson GL, Frost JW and Ober RE (1979) Human plasma pharmacokinetics of flecainide acetate (R818), a new antiarrhythmic following single oral and intravenous doses. Clin Pharmacol Ther 25: 218
Conard GJ and Ober RE (1984) Metabolism of flecainide. Am J Cardiol 53: 41B-51B
Tjandramaga TB, Verbesselt R, van Hecken A, Mullie A, Schepper PJ (1984) IV and oral flecainide kinetics: absolute bioavailability, effects of foods, antacid (aluminium hydroxide) and multiple oral doses. Eur Heart J 5 [Suppl B]: 135
Margolis B, DeSilva RA, Lown B (1980) Episodic drug treatment in the management of paroxysmal arrhythmias. Am J Cardiol 45: 621–626
Lie-A-Huen L, Stuurman RM, IJdenberg FN, Kingma JH, Meijer DKF (1989) High performance liquid chromatographic assay of flecainide and its enantiomers in serum. Ther Drug Monit 6: 708–712
Proost JH (1987) Critical evaluation of the determination of bioavailability by numerical deconvolution. Thesis, University of Groningen, The Netherlands
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lie-A-Huen, L., Proost, J.H., Kingma, J.H. et al. Absorption kinetics of oral and rectal flecainide in healthy subjects. Eur J Clin Pharmacol 38, 595–598 (1990). https://doi.org/10.1007/BF00278588
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00278588