Article Text
Abstract
Objective ACE inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) are widely prescribed in patients with high cardiovascular (CV) risk. However, whether both classes have equivalent effectiveness to prevent CV events remains unclear. The aim of this study was to compare the incidence of major CV events between ACEI and ARB users.
Methods The Reduction of Atherothrombosis for Continued Health registry is an observational study who enrolled 69 055 individuals with high CV risk. Among them, 40 625 patients (ACEIs 67.9% and ARBs 32.1%) were included. Main outcome was rates of CV mortality, non-fatal myocardial infarction, non-fatal stroke or hospitalisation for CV disease at 4 years.
Results In a propensity score-adjusted cohort, the incidence of the primary outcome was lower in patients on ARBs compared with ACEIs (29.2% vs 33.4%; adjusted HR 0.90; 95% CI 0.86 to 0.95; p<0.001). Similar results were observed for CV (6.9% vs 8.2%; HR 0.83; 95% CI 0.75 to 0.93; p=0.001) and all-cause mortality (11.6% vs 12.6%; HR 0.89; 95% CI 0.82 to 0.97; p=0.005). Analyses using propensity score matching yielded similar results. History of diabetes or estimated glomerular filtration rate did not affect the results. ARB use was associated with lower rates of all-cause mortality in secondary prevention but not in primary prevention patients (p-value for interaction=0.03).
Conclusion ARB use appears to be associated with 10% lower rates of CV events compared with ACEIs, especially in patients with established CV disease. Our results suggest that ARBs may provide superior protection against CV events than ACEIs in high-risk patients in real-world practice.
- Cardiac risk factors and prevention
- Epidemiology
- Coronary artery disease
Statistics from Altmetric.com
Introduction
ACE inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) are widely prescribed in patients with a high cardiovascular (CV) risk. Both classes target the renin–angiotensin system (RAS) but have different sites of action.1 Potential difference in comparative efficacy of ACEIs and ARBs is a subject of debate.2 Both American and European guidelines for the management of patient with CV disease recommend ACEIs as a first choice therapy, whereas ARBs are an alternative therapy for ACEI-intolerant patients.3 4 These recommendations are based on the marked benefit of ACEIs in randomised clinical trials (RCTs) compared with no consistent benefit of ARBs versus placebo in stable CV disease. However, a recent meta-analysis highlighted ARBs to be as efficacious and safe as ACEIs when analysis was restricted to trials after the year 2000, when aggressive prevention therapies were routinely used.2 Moreover, no difference between the two classes was observed in recent head-to-head RCTs of ACEIs versus ARBs,5–9 but these studies were limited to highly selected patients, and their extrapolation to the average patient may be debatable.10 Conversely, observational studies could provide a better external validity than RCTs to evaluate the effect of long-term use of drugs in real-world populations.11 12 For example, analyses from the Reduction of Atherothrombosis for Continued Health (REACH) cohort challenged the common views on efficacy of β-blockers or renin–angiotensin antagonists in subjects with stable coronary artery disease.13 14 Finally, recent meta-analyses that assessed efficacy of blood pressure–lowering agents on CV outcomes in patients with hypertension or diabetes reported conflicting results regarding comparisons between ACEIs and ARBs.15–18
The REACH registry provides an opportunity to directly compare ACEIs’ and ARBs’ effectiveness in routine clinical practice, in a contemporary large cohort of outpatients with high CV risk, with a variety of comorbidities, broad geographic representation and under well-defined intensity of preventive therapy. Data from this cohort were used to compare the incidence of mortality and CV events according to the use of ACEIs or ARBs.
Methods
Study population
The design, methods and primary results of the international REACH registry have been published previously.19 Briefly, 69 055 patients at least 45 years old with ≥3 risk factors for atherosclerosis and patients with previous CV disease were enrolled between 2003 and 2004. Risk factors taken into account included history of diabetes, diabetic nephropathy, ankle-brachial index ≤0.9, asymptomatic carotid stenosis of ≥70%, carotid intima media thickness at least two times that at adjacent sites, systolic blood pressure ≥150 mm Hg despite treatment, hypercholesterolaemia treated with medication, current smoking of ≥15 cigarettes per day and age ≥65 years for men or ≥70 years for women. Signed informed consent was obtained from all patients, and the institutional review board in each country approved the protocol according to the local requirements. These patients were assessed annually at years 1 through 4, and follow-up was completed in 2008. Patients included in the present analysis were those with complete 4-year follow-up information and ACEI or ARB status at baseline. Patients taking a combination of ACEI and ARB were excluded from the analysis (figure 1).
Flow diagram. ACEI, ACE inhibitor; ARB, angiotensin receptor blocker; REACH, Reduction of Atherothrombosis for Continued Health.
Outcomes measures
The primary outcome was the composite of CV mortality, non-fatal myocardial infarction (MI), non-fatal stroke or hospitalisation for CV reasons during the 4-year follow-up. Individual components of the primary outcome, all-cause mortality and heart failure (HF) (defined by signs or symptoms of left or right ventricular failure or both) were also studied.
Statistical analysis
Continuous variables are expressed as mean (SD), and categorical variables are expressed as frequencies and percentages. To account for differences in baseline characteristics between treatment groups (see online supplementary table 1), propensity score analysis was performed to further account for these differences between ACEIs and ARBs users.
Supplementary file
To calculate propensity score, 10 041 patients were excluded due to missing data in covariates used to calculate the propensity score. No major difference was observed between patients included and excluded to calculate propensity score (see online supplementary table 2). Propensity scores for all patients were first estimated using a non-parsimonious multivariable logistic regression model, with ACEIs or ARBs use as the dependent variable and 17 baseline characteristics as covariates (sex, age, estimated glomerular filtration rate (eGFR), geographical regions (divided in two groups), current smoker status, hypertension, history of HF, history of atrial fibrillation, history of coronary artery disease, history of MI, history of diabetes, aspirin use, statin use, beta blockers use, calcium channel blockers use, nitrates use and diuretics use).
Cox models for primary and secondary outcomes were conducted with ACEI versus ARB status as covariate after adjustment on propensity score and an additional analysis was performed by matching ARBs users with ACEIs users, based on their propensity score. The matching was performed by a SAS macro20 implementing an algorithm based on 8 to 1 digit matching (ratio of 1:1 without replacement). For ACEI user, ARB user with similar propensity scores was selected. This matched couple was thus removed from the cohort, and the matching procedure was iteratively rerun until a maximum number of patient couples were assembled. We were able to match 19 804 patients, of which 9902 patients on ARBs (76% of the ARB cohort) and 9902 patients on ACEIs (35.9% of the ACEI cohort).
Absolute standardised differences for all covariates before and after matching were estimated to evaluate bias reduction using the propensity score matching method. After propensity score matching, all absolute standardised differences were <10%, indicating robust matching (see online supplementary figure 1). Statistical tests of comparisons in baseline characteristics between the matched groups were also non-significant (p>0.05) (see online supplementary table 3).
In the propensity score-adjusted cohort, Cox proportional hazard regression model with the propensity score introduced as a covariate was used to compare the risk of outcomes between both groups. In the propensity score-matched cohort, we used cox proportional hazard regression model stratified on the matched pairs.
Subgroup analyses were also performed in the propensity score-adjusted and score-matched cohorts according to diabetes, eGFR values (divided into three groups: ≤30, >30 and ≤60, >60 mL/min), established arterial disease and HF at baseline. We also compared ACEIs and ARBs according to the numbers of medication use in addition to ACEIs or ARBs and believed to reduce the incidence of CV events: optimal medical therapy, that is, statins, β-blockers and antiplatelet agents, including aspirin.
Heterogeneity across subgroups was tested by introducing interaction terms into the Cox proportional hazard regression models.
Proportional hazard assumptions were checked by testing the interaction between ACEI versus ARB status and log (time) within the model.
Analysis was performed using SAS version 9.3 (SAS Institute, Cary, North Carolina).
Results
Study population
From the 68 236 patients with available baseline data, 65 531 were enrolled for 4-year follow-up. Among them, 40 625 patients (70.0%) had ACEI or ARB user status. Patients were divided into two groups according to ACEI or ARB use at baseline (27 589 (67.9%) ACEI-only users, 13 036 (32.1%) ARBonly users) (figure 1).
Among those patients who used ACEI or ARB at baseline, discontinuation was similar: the proportion of patients remaining on ACEIs or ARBs was 88.3% and 87.7% at 12 months, 84.9% and 84.5% at 2 years, 80.0% and 81.5% at 3 years, and 76.8% and 78.8% at 4 years, respectively.
Study outcomes
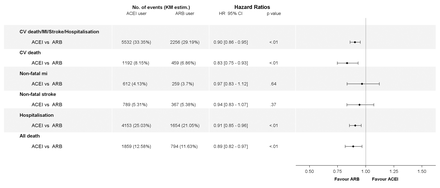
In the propensity score-adjusted cohort, at least one of the components of the primary outcome occurred in 7788 patients during the 4-year follow up (cumulative incidence rate of 32.01%). Compared with ACEI users, the rate of the primary outcome was lower in ARB users (adjusted HR 0.90; 95% CI 0.86 to 0.95; p<0.001) (figure 2). The individual components of the primary endpoint and all-cause mortality were lower in ARBs users for CV mortality (HR 0.83; 95% CI 0.75 to 0.93; p=0.001), hospitalisation for CV reasons (HR 0.91; 95% CI 0.85 to 0.96; p<0.001) and all-cause mortality (HR 0.89; 95% CI 0.82 to 0.97; p=0.005). There was no difference in the other components of the primary outcome (non-fatal MI, non-fatal stroke) in the propensity score-adjusted cohort. HF occurred in 2473 patients during the 4-year follow up and was similar between the two groups (8.49% and 7.24% in ACEI and ARB groups, respectively; HR 0.96, 95% CI 0.88 to 1.06; p=0.418).
Risks of outcomes in ACEIs versus ARBs cohorts, in propensity score-adjusted analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blockers; CV, cardiovascular; MI, myocardial infarction.
Similar results to the propensity score-matched cohort were observed. The adjusted HR was 0.91 (95% CI 0.85 to 0.97; p=0.005) for the primary outcome, 0.80 (95% CI 0.69 to 0.92; p=0.001) for C mortality and 0.84 (95% CI 0.75 to 0.94; p=0.002) for all-cause mortality (figure 3).
Risks of outcomes in ACEIs versus ARBs cohorts, in propensity score-matched analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blocker; CV, cardiovascular; MI, myocardial infarction.
Subgroup analysis
In both cohorts, history of diabetes at baseline or level of eGFR did not affect the primary results. Lower rates of primary outcome, CV mortality, hospitalisation for CV reasons or all-cause mortality were observed in ARB users compared with ACEI users (figures 4–7). However, in patients without established atherosclerosis (primary prevention), there was no difference between ARB or ACEI users for all outcomes, whereas in established atherosclerosis (secondary prevention) ARB use was associated with a reduction of the primary outcome, CV mortality, hospitalisation for CV reasons or all-cause mortality, although the interaction was significant only for CV mortality in the propensity score-matched and adjusted cohort (p value for interaction=0.03 in both cohorts).
Risks of outcomes (primary outcomes, CV death, Non-fatal MI) in ACEIs versus ARBs cohorts in subgroups, in propensity score-adjusted analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blocker; CV, cardiovascular; MI, myocardial infarction; HF, heart failure; GFR, glomerular filtration rate.
Risks of outcomes (non-fatal stroke, hospitalisation, all death) in ACEIs versus ARBs cohorts in subgroups, in propensity score-adjusted analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blocker; CV, cardiovascular; MI, myocardial infarction; HF, heart failure; GFR, glomerular filtration rate.
Risks of outcomes (primary outcomes, CV death, non-fatal MI) in ACEIs versus ARBs cohorts in several subgroups, in propensity score-matched analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blocker; CV, cardiovascular; MI, myocardial infarction; HF, heart failure; GFR, glomerular filtration rate.
Risks of outcomes (non-fatal stroke, hospitalisation, all death) in ACEIs versus ARBs cohorts in several subgroups, in propensity score-matched analysis. ACEIs, ACE inhibitors; ARB, angiotensin receptor blocker; CV, cardiovascular; MI, myocardial infarction; HF, heart failure; GFR, glomerular filtration rate.
Similarly, in both cohorts, ARB use was associated with a lower rate of non-fatal MI in patients with history of HF but not in patients without HF (figures 4–7). This effect of ARBs or ACEIs for non-fatal MI interacted significantly in both cohorts (p value for interaction=0.04 and 0.01 in propensity score-adjusted and matched cohorts, respectively). Otherwise, history of HF at baseline did not affect the similar rate of HF during follow-up in patients taking ACEIs and ARBs (data not shown).
We compared ACEIs and ARBs according to the use of optimal medical therapy for the prevention of CV disease. As expected, CV or all-cause mortality decreased with the number of treatments used (see online supplementary figures 2, 3) in both cohorts. In the propensity score-adjusted cohort, the difference between ACEIs and ARBs for primary outcomes, CV and all-cause mortality was gradually attenuated with the optimisation of CV treatments. This attenuation was significant for CV and all-cause mortality (p value for interaction <0.01 in both parameters).
Discussion
In this analysis of 40 625 people from the REACH registry, we found that, in comparison with ACEI use, routine use of ARBs was associated with a lower rate of major CV events in outpatients with high CV risk. We observed a consistency of our findings after multiple adjustment methods. Moreover, subgroup analysis found similar results regardless of a history of diabetes, HF or chronic kidney disease. However, the difference between ARB and ACEI use was only significant in the subgroup of patients with established atherosclerosis, with a significant interaction for all-cause mortality.
In most RCTs, ACEIs and ARBs have been associated with decreased major CV events compared with placebo or active comparators, including in patients with high CV risk.21–24 However, due to the different mechanisms of action of these two classes of drugs, differential clinical effect could be expected.1 Indeed, in the meta-analysis of Van Vark et al, treatment with ACEIs, but not ARBs, reduced mortality in patients with hypertension.15 Likewise, a recent network meta-analysis concluded that ACEIs reduced all-cause mortality, CV mortality and major CV events, whereas ARBs had no benefits on these outcomes in patients with diabetes.16 However, a recent meta-analysis pointed out several differences in the ACEIs versus placebo and ARBs versus placebo trials.2 One was that most of trials with ACEIs were conducted a decade earlier than the ARB trials with a lower placebo event rate, which reflected the concomitant optimised medication use in the ARB trials versus the ACEI trials. In that report, when analyses were restricted to trials published after the year 2000, outcomes were similar between ACEIs versus placebo and ARBs versus placebo. In line with these results, when we analysed the interaction between ACEI/ARB and quality of therapy, we found a significant attenuation of the beneficial effect of ARBs versus ACEIs on mortality with the optimisation of medical therapy.
In the few head-to-head RCTs that compared ACEIs and ARBs, no difference was found between both classes. However, it is noteworthy that in the three oldest studies, doses of ARB used were lower than the higher dose recommended.5 25 26 Moreover, the neutral results of the three recent head-to-head trials may be a consequence of a more aggressive use of a background optimal medical therapy, as concomitantly recommended.6–8 Another difference in ACEIs and ARBs trials was that more patients with a history of coronary or other vascular disease were included in ACEI trials than in ARB trials.2 These differences could also explain discrepancies between these previous meta-analyses and our results, suggesting that ARBs could be more beneficial than ACEIs in patients with a prior history of CV disease. Furthermore, a recent meta-analysis of the association between blood pressure–lowering treatments and vascular disease in patients with type 2 diabetes showed that ARBs were associated with a significant reduction in mortality compared with any active comparator including ACEIs.17 A similar trend was observed with ARBs, but not with ACEIs, on all-cause mortality in patients with diabetes and kidney disease in the network meta-analysis of Palmer et al.18 It is noteworthy that in most of these meta-analyses, moderate or higher heterogeneity between populations included in trials limited the validity of the pooled analysis. Moreover, results from RCTs comparing ACEIs or ARBs with active controls showed that the blood pressure–lowering independent effect of both classes are less evident in patients without previous CV disease than in secondary CV prevention population.2 This observation could explain the lack of difference between ACEI and ARB use observed in our primary prevention population.
In contrast to our results on primary outcomes and all-cause mortality, lack of difference on incident HF between the two groups in the whole population and in subgroups of patients with or without HF at baseline was surprising. Moreover, as mentioned in the 2016 ESC guidelines on the management of patients with HF, ARBs are recommended only as an alternative in patients intolerant of an ACEI.27 This recommendation is based on the fact that there are more RCTs supporting the use of ACEIs than of ARBs. However, our findings were in line with previous head-to-head trials in patients with or without HF at baseline in which no difference on HF between both classes was observed.6–8 26
It would have been interesting to study the effect of a combination of ARBs and ACEIs at baseline. Unfortunately, the small size of this subgroup (1571 patients) did not allow to perform any convincing propensity score–based analysis.
Despite our findings are derived from a large observational cohort with a 4-year follow-up in a broad range populations, our analysis has limitations. Although propensity score adjustment, the potential for residual confounding by indication due to unmeasured or measured confounders, cannot be completely ruled out. However, reproducibility of the results across several models, subgroups and adjustment methods used suggest that these results are valid. Moreover, the size of the population and the attendant narrow CIs of clinical outcome rates reinforce the strength of our findings. Another important limitation was we did not have data regarding doses of ACEIs and ARBs used. It is well known that the beneficial effect of RAS antagonists observed in RCTs is obtained from using higher doses of ACEIs or ARBs21 28–30 whereas doses used in routine clinical practice are often lower. Likewise, this study cannot take into account the differences that may exist between drugs within the same class due to the lack of this information.
In conclusion, in the large international REACH registry, ARB use appears associated with lower rates of major CV outcomes compared with ACEI use, especially in patients with established CV disease. Despite a greater level of evidence for ACEIs than for ARBs from RCTs and a similar effect of both classes in head-to-head RCTs, these results question the differential effect of ACEIs and ARBs in patients in secondary prevention in actual clinical practice.
Key messages
What is already known on this subject?
Both American and European guidelines for the management of patient with cardiovascular disease recommend ACE inhibitors (ACEIs) as a first-choice therapy whereas angiotensin receptor blockers (ARBs) are an alternative therapy for ACEI-intolerant patients. However, data available from RCTs and meta-analyses reported conflicting results regarding comparisons between ACEIs and ARBs. Whether ACEIs and ARBs have equivalent effectiveness to prevent cardiovascular events remains unclear.
What might this study add?
Our results from a contemporary large cohort of outpatients with high cardiovascular risk show that the incidence of major adverse cardiovascular events was significantly lower in patients on ARBs compared with ACEIs (29.2% vs 33.4%; adjusted HR 0.90; 95% CI 0.86 to 0.95; p<0.001), especially in patients with established cardiovascular disease.
How might this impact on clinical practice?
Our results suggest that ARBs should be preferred to ACEIs in patients with a previous history of cardiovascular diseases.
References
Footnotes
Contributors Drafting the work: LP, RR, YE; Revising the work critically for important intellectual content: YE, MM, UZ, CMR, EMO, KAE, DLB, PGS; Substantial contributions to the conception or design of the work, and the acquisition of data: UZ, CMR, EMO, KAE, DLB and PGS; Substantial contributions to analysis or interpretation of data: LP, RR, YE, MM, PGS; Final approval of the version published: LP, RR, YE, MM, UZ, CMR, EMO, KAE, DLB, PGS.
Funding The REACH Registry is endorsed by the World Heart Federation. The REACH Registry was supported by Sanofi-Aventis, Bristol-Myers Squibb, and the Waksman Foundation (Tokyo, Japan).
Competing interests Dr LP reports grants and personal fees from Sanofi, personal fees from Servier, outside the
submitted work; Dr RR reports grants and personal fees from Sanofi, personal fees from
AstraZeneca, personal fees and non-financial support from Novartis, personal fees from
MSD, outside the submitted work; M. YE has nothing to disclose; Dr MM reports grants
and personal fees from MSD, Novartis, Novo Nordisk, Sanofi and Servier, personal fees and
non-financial support from Abbott, Intarcia, and Eli Lilly, outside the submitted work; Dr
UZ reports personal fees from Astra Zeneca, personal fees from Boehringer Ingelheim,
personal fees from Novartis, personal fees from BMS, personal fees from Sanofi, personal
fees from MSD, personal fees from Pfizer, outside the submitted work; Dr. Reid reports
grants from Sanofi Aventis, during the conduct of the study; Dr MO reports personal fees
from Abbott Vascular, personal fees from Abiomed, personal fees from Astra Zeneca, personal fees from Biotie, personal fees from Boehringer Inelheim, personal fees from Bristol Myers Squibb, grants and personal fees from Daiichi Sankyo, personal fees from Faculty
Connection, grants from Gilead Sciences, grants and personal fees from Janssen Parmaceuticals, personal fees from Merck, personal fees from Sanofi Aventis, personal fees
from St. Jude Medical, personal fees from Stealth eptides, personal fees from The Medicines
Company, personal fees from Medscape, outside the submitted work; Dr KAE has nothing
to disclose; D.L.B. reports grants from Bristol Myers Squibb, grants from Sanofi Aventis,
during the conduct of the study; grants from Amarin, grants from AstraZeneca, grants from
Bristol-Myers Squibb, grants from Eisai, grants from Ethicon, grants from Medtronic, grants
from sanofi aventis, grants from The Medicines Company, other from FlowCo, other from
PLx Pharma, other from Takeda, personal fees from Duke Clinical Research Institute,
Personal fees from Mayo Clinic, personal fees from Population Health Research Institute,
personal fees and non-financial support from American College of Cardiology, personal fees from Belvoir Publications, personal fees from Slack Publications, personal fees from
WebMD, personal fees from Elsevier, other from Medscape Cardiology, other from Regado
Biosciences, other from Boston VA Research Institute, personal fees and non-financial
support from Society of Cardiovascular Patient Care, non-financial support from American Heart Association, personal fees from HMP Communications, grants from Roche, personal fees from Harvard Clinical Research Institute, other from Clinical Cardiology, personal fees from Journal of the American College of Cardiology,
other from VA, grants from Pfizer, grants from Forest Laboratories, grants from Ischemix,
other from St. Jude Medical, other from Biotronik, other from Cardax, other from Ameri
can College of Cardiology, other from Boston Scientific, grants from Amgen, outside the
submitted work; Dr PG reports personal fees from BMS and Sanofi, during the conduct of the
study; grants from Merck, Sanofi, and Servier, personal fees from Amarin, AstraZeneca, Ba
yer, Boehringer-Ingelheim, Bristol-Myers-Squibb, CSL-Behring, Daiichi-Sankyo, GlaxoSmithKline, Janssen, Lilly, Merck Novartis, Pfizer, Regeneron, Sanofi, Servier, The M
edicines Company, outside the submitted work.
Provenance and peer review Not commissioned; externally peer reviewed.