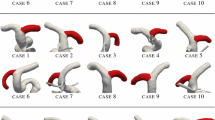
Abstract
Cardiovascular pathologies such as intracranial aneurysms (IAs) and atherosclerosis preferentially localize to bifurcations and curvatures where hemodynamics are complex. While extensive knowledge about low wall shear stress (WSS) has been generated in the past, due to its strong relevance to atherogenesis, high WSS (typically >3 Pa) has emerged as a key regulator of vascular biology and pathology as well, receiving renewed interests. As reviewed here, chronic high WSS not only stimulates adaptive outward remodeling, but also contributes to saccular IA formation (at bifurcation apices or outer curves) and atherosclerotic plaque destabilization (in stenosed vessels). Recent advances in understanding IA pathogenesis have shed new light on the role of high WSS in pathological vascular remodeling. In complex geometries, high WSS can couple with significant spatial WSS gradient (WSSG). A combination of high WSS and positive WSSG has been shown to trigger aneurysm initiation. Since endothelial cells (ECs) are sensors of WSS, we have begun to elucidate EC responses to high WSS alone and in combination with WSSG. Understanding such responses will provide insight into not only aneurysm formation, but also plaque destabilization and other vascular pathologies and potentially lead to improved strategies for disease management and novel targets for pharmacological intervention.






Similar content being viewed by others
References
Abruzzo, T., A. Kendler, R. Apkarian, M. Workman, J. C. Khoury, and H. J. Cloft. Cerebral aneurysm formation in nitric oxide synthase-3 knockout mice. Curr. Neurovasc. Res. 4:161–169, 2007.
Alnaes, M. S., J. Isaksen, K. A. Mardal, B. Romner, M. K. Morgan, and T. Ingebrigtsen. Computation of hemodynamics in the circle of Willis. Stroke 38:2500–2505, 2007.
Aoki, T., H. Kataoka, R. Ishibashi, K. Nozaki, K. Egashira, and N. Hashimoto. Impact of monocyte chemoattractant protein-1 deficiency on cerebral aneurysm formation. Stroke 40:942–951, 2009.
Aoki, T., H. Kataoka, R. Ishibashi, K. Nozaki, and N. Hashimoto. Cathepsin B, K, and S are expressed in cerebral aneurysms and promote the progression of cerebral aneurysms. Stroke 39:2603–2610, 2008.
Aoki, T., H. Kataoka, M. Morimoto, K. Nozaki, and N. Hashimoto. Macrophage-derived matrix metalloproteinase-2 and -9 promote the progression of cerebral aneurysms in rats. Stroke 38:162–169, 2007.
Aoki, T., H. Kataoka, M. Nishimura, R. Ishibashi, R. Morishita, and S. Miyamoto. Ets-1 promotes the progression of cerebral aneurysm by inducing the expression of MCP-1 in vascular smooth muscle cells. Gene Ther. 17:1117–1123, 2010.
Aoki, T., H. Kataoka, M. Shimamura, H. Nakagami, K. Wakayama, T. Moriwaki, R. Ishibashi, K. Nozaki, R. Morishita, and N. Hashimoto. NF-kappaB is a key mediator of cerebral aneurysm formation. Circulation 116:2830–2840, 2007.
Aoki, T., M. Nishimura, H. Kataoka, R. Ishibashi, K. Nozaki, and S. Miyamoto. Complementary inhibition of cerebral aneurysm formation by eNOS and nNOS. Lab. Invest. 91:619–626, 2011.
Aoki, T., M. Nishimura, T. Matsuoka, K. Yamamoto, T. Furuyashiki, H. Kataoka, S. Kitaoka, R. Ishibashi, A. Ishibazawa, S. Miyamoto, R. Morishita, J. Ando, N. Hashimoto, K. Nozaki, and S. Narumiya. PGE(2) -EP(2) signalling in endothelium is activated by haemodynamic stress and induces cerebral aneurysm through an amplifying loop via NF-kappaB. Br. J. Pharmacol. 163:1237–1249, 2011.
Bark, Jr., D. L., A. N. Para, and D. N. Ku. Correlation of thrombosis growth rate to pathological wall shear rate during platelet accumulation. Biotechnol. Bioeng. 109:2642–2650, 2012.
Brown, Jr., R. D., D. O. Wiebers, and G. S. Forbes. Unruptured intracranial aneurysms and arteriovenous malformations: frequency of intracranial hemorrhage and relationship of lesions. J. Neurosurg. 73:859–863, 1990.
Bruno, G., R. Todor, I. Lewis, and D. Chyatte. Vascular extracellular matrix remodeling in cerebral aneurysms. J. Neurosurg. 89:431–440, 1998.
Burke, A., and G. A. Fitzgerald. Oxidative stress and smoking-induced vascular injury. Prog. Cardiovasc. Dis. 46:79–90, 2003.
Caro, C. G., J. M. Fitz-Gerald, and R. C. Schroter. Atheroma and arterial wall shear. Observation, correlation and proposal of a shear dependent mass transfer mechanism for atherogenesis. Proc. R. Soc. Lond. B Biol. Sci. 177:109–159, 1971.
Castier, Y., R. P. Brandes, G. Leseche, A. Tedgui, and S. Lehoux. p47phox-dependent NADPH oxidase regulates flow-induced vascular remodeling. Circ. Res. 97:533–540, 2005.
Chatzizisis, Y. S., M. Jonas, A. U. Coskun, R. Beigel, B. V. Stone, C. Maynard, R. G. Gerrity, W. Daley, C. Rogers, E. R. Edelman, C. L. Feldman, and P. H. Stone. Prediction of the localization of high-risk coronary atherosclerotic plaques on the basis of low endothelial shear stress: an intravascular ultrasound and histopathology natural history study. Circulation 117:993–1002, 2008.
Chien, S. Mechanotransduction and endothelial cell homeostasis: the wisdom of the cell. Am. J. Physiol. Heart Circ. Physiol. 292:H1209–H1224, 2007.
Davies, P. F. Flow-mediated endothelial mechanotransduction. Physiol. Rev. 75:519–560, 1995.
DePaola, N., P. F. Davies, W. F. Pritchard, Jr., L. Florez, N. Harbeck, and D. C. Polacek. Spatial and temporal regulation of gap junction connexin43 in vascular endothelial cells exposed to controlled disturbed flows in vitro. Proc. Natl Acad. Sci. USA 96:3154–3159, 1999.
Dolan, J. M., H. Meng, S. Singh, R. Paluch, and J. Kolega. High fluid shear stress and spatial shear stress gradients affect endothelial proliferation, survival, and alignment. Ann. Biomed. Eng. 39:1620–1631, 2011.
Dolan, J. M., F. J. Sim, H. Meng, and J. Kolega. Endothelial cells express a unique transcriptional profile under very high wall shear stress known to induce expansive arterial remodeling. Am. J. Physiol. Cell Physiol. 302:C1109–C1118, 2012.
Dumont, O., L. Loufrani, and D. Henrion. Key role of the NO-pathway and matrix metalloprotease-9 in high blood flow-induced remodeling of rat resistance arteries. Arterioscler. Thromb. Vasc. Biol. 27:317–324, 2007.
Eldawoody, H., H. Shimizu, N. Kimura, A. Saito, T. Nakayama, A. Takahashi, and T. Tominaga. Simplified experimental cerebral aneurysm model in rats: comprehensive evaluation of induced aneurysms and arterial changes in the circle of Willis. Brain Res. 1300:159–168, 2009.
Frosen, J., R. Tulamo, A. Paetau, E. Laaksamo, M. Korja, A. Laakso, M. Niemela, and J. Hernesniemi. Saccular intracranial aneurysm: pathology and mechanisms. Acta Neuropathol. 123:773–786, 2012.
Fujii, K., Y. Kobayashi, G. S. Mintz, H. Takebayashi, G. Dangas, I. Moussa, R. Mehran, A. J. Lansky, E. Kreps, M. Collins, A. Colombo, G. W. Stone, M. B. Leon, and J. W. Moses. Intravascular ultrasound assessment of ulcerated ruptured plaques: a comparison of culprit and nonculprit lesions of patients with acute coronary syndromes and lesions in patients without acute coronary syndromes. Circulation 108:2473–2478, 2003.
Fukuda, S., N. Hashimoto, H. Naritomi, I. Nagata, K. Nozaki, S. Kondo, M. Kurino, and H. Kikuchi. Prevention of rat cerebral aneurysm formation by inhibition of nitric oxide synthase. Circulation 101:2532–2538, 2000.
Gao, L., Y. Hoi, D. D. Swartz, J. Kolega, A. Siddiqui, and H. Meng. Nascent aneurysm formation at the basilar terminus induced by hemodynamics. Stroke 39:2085–2090, 2008.
Gertz, S. D., and W. C. Roberts. Hemodynamic shear force in rupture of coronary arterial atherosclerotic plaques. Am. J. Cardiol. 66:1368–1372, 1990.
Gibbons, G. H., and V. J. Dzau. The emerging concept of vascular remodeling. N. Engl. J. Med. 330:1431–1438, 1994.
Gijsen, F. J., F. Mastik, J. A. Schaar, J. C. Schuurbiers, W. J. van der Giessen, P. J. de Feyter, P. W. Serruys, A. F. van der Steen, and J. J. Wentzel. High shear stress induces a strain increase in human coronary plaques over a 6-month period. EuroIntervention. 7:121–127, 2011.
Gijsen, F. J., J. J. Wentzel, A. Thury, F. Mastik, J. A. Schaar, J. C. Schuurbiers, C. J. Slager, W. J. van der Giessen, P. J. de Feyter, A. F. van der Steen, and P. W. Serruys. Strain distribution over plaques in human coronary arteries relates to shear stress. Am. J. Physiol. Heart Circ. Physiol. 295:H1608–H1614, 2008.
Glagov, S., E. Weisenberg, C. K. Zarins, R. Stankunavicius, and G. J. Kolettis. Compensatory enlargement of human atherosclerotic coronary arteries. N. Engl. J. Med. 316:1371–1375, 1987.
Glagov, S., C. Zarins, D. P. Giddens, and D. N. Ku. Hemodynamics and atherosclerosis. Insights and perspectives gained from studies of human arteries. Arch. Pathol. Lab. Med. 112:1018–1031, 1988.
Greve, J. M., A. S. Les, B. T. Tang, M. T. Draney Blomme, N. M. Wilson, R. L. Dalman, N. J. Pelc, and C. A. Taylor. Allometric scaling of wall shear stress from mice to humans: quantification using cine phase-contrast MRI and computational fluid dynamics. Am. J. Physiol. Heart Circ. Physiol. 291:H1700–H1708, 2006.
Groen, H. C., F. J. Gijsen, A. van der Lugt, M. S. Ferguson, T. S. Hatsukami, A. F. van der Steen, C. Yuan, and J. J. Wentzel. Plaque rupture in the carotid artery is localized at the high shear stress region: a case report. Stroke 38:2379–2381, 2007.
Guzman, R. J., K. Abe, and C. K. Zarins. Flow-induced arterial enlargement is inhibited by suppression of nitric oxide synthase activity in vivo. Surgery 122:273–279, 1997; discussion 279–280.
Han, H. C. Twisted blood vessels: symptoms, etiology and biomechanical mechanisms. J. Vasc. Res. 49:185–197, 2012.
Hashimoto, N., H. Handa, and F. Hazama. Experimentally induced cerebral aneurysms in rats. Surg. Neurol. 10:3–8, 1978.
Hashimoto, N., H. Handa, I. Nagata, and F. Hazama. Experimentally induced cerebral aneurysms in rats: Part V. Relation of hemodynamics in the circle of Willis to formation of aneurysms. Surg. Neurol. 13:41–45, 1980.
Hashimoto, T., H. Meng, and W. L. Young. Intracranial aneurysms: links among inflammation, hemodynamics and vascular remodeling. Neurol. Res. 28:372–380, 2006.
Hazama, F., H. Kataoka, E. Yamada, K. Kayembe, N. Hashimoto, M. Kojima, and C. Kim. Early changes of experimentally induced cerebral aneurysms in rats. Light-microscopic study. Am. J. Pathol. 124:399–404, 1986.
Hoi, Y., L. Gao, M. Tremmel, R. A. Paluch, A. H. Siddiqui, H. Meng, and J. Mocco. In vivo assessment of rapid cerebrovascular morphological adaptation following acute blood flow increase. J. Neurosurg. 109:1141–1147, 2008.
Hsieh, H. J., N. Q. Li, and J. A. Frangos. Shear-induced platelet-derived growth factor gene expression in human endothelial cells is mediated by protein kinase C. J. Cell. Physiol. 150:552–558, 1992.
Hyun, S., C. Kleinstreuer, and J. P. Archie, Jr. Hemodynamics analyses of arterial expansions with implications to thrombosis and restenosis. Med. Eng. Phys. 22:13–27, 2000.
Ishibashi, A., Y. Yokokura, K. Kojima, and T. Abe. Acute obstructive hydrocephalus due to an unruptured basilar bifurcation aneurysm associated with bilateral internal carotid occlusion—a case report. Kurume Med. J. 40:21–25, 1993.
Jamous, M. A., S. Nagahiro, K. T. Kitazato, J. Satomi, and K. Satoh. Role of estrogen deficiency in the formation and progression of cerebral aneurysms. Part I: experimental study of the effect of oophorectomy in rats. J. Neurosurg. 103:1046–1051, 2005.
Jamous, M. A., S. Nagahiro, K. T. Kitazato, T. Tamura, H. A. Aziz, M. Shono, and K. Satoh. Endothelial injury and inflammatory response induced by hemodynamic changes preceding intracranial aneurysm formation: experimental study in rats. J. Neurosurg. 107:405–411, 2007.
Jesty, J., W. Yin, P. Perrotta, and D. Bluestein. Platelet activation in a circulating flow loop: combined effects of shear stress and exposure time. Platelets 14:143–149, 2003.
Jou, L. D., R. van Tyen, S. A. Berger, and D. Saloner. Calculation of the magnetization distribution for fluid flow in curved vessels. Magn. Reson. Med. 35:577–584, 1996.
Juvela, S. Natural history of unruptured intracranial aneurysms: risks for aneurysm formation, growth, and rupture. Acta Neurochir. Suppl. 82:27–30, 2002.
Kamiya, A., R. Bukhari, and T. Togawa. Adaptive regulation of wall shear stress optimizing vascular tree function. Bull. Math. Biol. 46:127–137, 1984.
Kamiya, A., and T. Togawa. Adaptive regulation of wall shear stress to flow change in the canine carotid artery. Am. J. Physiol. 239:H14–H21, 1980.
Karwowski, J. K., A. Markezich, J. Whitson, T. A. Abbruzzese, C. K. Zarins, and R. L. Dalman. Dose-dependent limitation of arterial enlargement by the matrix metalloproteinase inhibitor RS-113,456. J. Surg. Res. 87:122–129, 1999.
Kataoka, K., M. Taneda, T. Asai, A. Kinoshita, M. Ito, and R. Kuroda. Structural fragility and inflammatory response of ruptured cerebral aneurysms. A comparative study between ruptured and unruptured cerebral aneurysms. Stroke 30:1396–1401, 1999.
Kojima, M., H. Handa, N. Hashimoto, C. Kim, and F. Hazama. Early changes of experimentally induced cerebral aneurysms in rats: scanning electron microscopic study. Stroke 17:835–841, 1986.
Kolega, J., L. Gao, M. Mandelbaum, J. Mocco, A. H. Siddiqui, S. K. Natarajan, and H. Meng. Cellular and molecular responses of the basilar terminus to hemodynamics during intracranial aneurysm initiation in a rabbit model. J. Vasc. Res. 48:429–442, 2011.
Koskinas, K. C., C. L. Feldman, Y. S. Chatzizisis, A. U. Coskun, M. Jonas, C. Maynard, A. B. Baker, M. I. Papafaklis, E. R. Edelman, and P. H. Stone. Natural history of experimental coronary atherosclerosis and vascular remodeling in relation to endothelial shear stress: a serial, in vivo intravascular ultrasound study. Circulation 121:2092–2101, 2010.
Kulcsar, Z., A. Ugron, M. Marosfoi, Z. Berentei, G. Paal, and I. Szikora. Hemodynamics of cerebral aneurysm initiation: the role of wall shear stress and spatial wall shear stress gradient. AJNR Am. J. Neuroradiol. 32:587–594, 2011.
Kwak, B. R., P. Silacci, N. Stergiopulos, D. Hayoz, and P. Meda. Shear stress and cyclic circumferential stretch, but not pressure, alter connexin43 expression in endothelial cells. Cell Commun. Adhes. 12:261–270, 2005.
LaBarbera, M. Principles of design of fluid transport systems in zoology. Science 249:992–1000, 1990.
LaMack, J. A., and M. H. Friedman. Individual and combined effects of shear stress magnitude and spatial gradient on endothelial cell gene expression. Am. J. Physiol. Heart Circ. Physiol. 293:H2853–H2859, 2007.
Langille, B. L., and F. O’Donnell. Reductions in arterial diameter produced by chronic decreases in blood flow are endothelium-dependent. Science 231:405–407, 1986.
Leach, J. R., V. L. Rayz, B. Soares, M. Wintermark, M. R. Mofrad, and D. Saloner. Carotid atheroma rupture observed in vivo and FSI-predicted stress distribution based on pre-rupture imaging. Ann. Biomed. Eng. 38:2748–2765, 2010.
Lehman, R. M., G. K. Owens, N. F. Kassell, and K. Hongo. Mechanism of enlargement of major cerebral collateral arteries in rabbits. Stroke 22:499–504, 1991.
Lehoux, S., Y. Castier, and A. Tedgui. Molecular mechanisms of the vascular responses to haemodynamic forces. J. Intern. Med. 259:381–392, 2006.
Lehoux, S., F. Tronc, and A. Tedgui. Mechanisms of blood flow-induced vascular enlargement. Biorheology. 39:319–324, 2002.
Levkau, B., R. D. Kenagy, A. Karsan, B. Weitkamp, A. W. Clowes, R. Ross, and E. W. Raines. Activation of metalloproteinases and their association with integrins: an auxiliary apoptotic pathway in human endothelial cells. Cell Death Differ. 9:1360–1367, 2002.
Li, Z. Y., V. Taviani, T. Tang, U. Sadat, V. Young, A. Patterson, M. Graves, and J. H. Gillard. The mechanical triggers of plaque rupture: shear stress vs pressure gradient. Br. J. Radiol. 82(Spec No. 1):S39–S45, 2009.
Lindekleiv, H. M., K. Valen-Sendstad, M. K. Morgan, K. A. Mardal, K. Faulder, J. H. Magnus, K. Waterloo, B. Romner, and T. Ingebrigtsen. Sex differences in intracranial arterial bifurcations. Gend. Med. 7:149–155, 2010.
Lovett, J. K., and P. M. Rothwell. Site of carotid plaque ulceration in relation to direction of blood flow: an angiographic and pathological study. Cerebrovasc. Dis. 16:369–375, 2003.
Malek, A. M., S. L. Alper, and S. Izumo. Hemodynamic shear stress and its role in atherosclerosis. JAMA 282:2035–2042, 1999.
Malek, A. M., G. H. Gibbons, V. J. Dzau, and S. Izumo. Fluid shear stress differentially modulates expression of genes encoding basic fibroblast growth factor and platelet-derived growth factor B chain in vascular endothelium. J. Clin. Invest. 92:2013–2021, 1993.
Masuda, H., Y. J. Zhuang, T. M. Singh, K. Kawamura, M. Murakami, C. K. Zarins, and S. Glagov. Adaptive remodeling of internal elastic lamina and endothelial lining during flow-induced arterial enlargement. Arterioscler. Thromb. Vasc. Biol. 19:2298–2307, 1999.
Meng, H., E. Metaxa, L. Gao, N. Liaw, S. K. Natarajan, D. D. Swartz, A. H. Siddiqui, J. Kolega, and J. Mocco. Progressive aneurysm development following hemodynamic insult. J. Neurosurg. 114:1095–1103, 2011.
Meng, H., D. D. Swartz, Z. Wang, Y. Hoi, J. Kolega, E. M. Metaxa, M. P. Szymanski, J. Yamamoto, E. Sauvageau, and E. I. Levy. A model system for mapping vascular responses to complex hemodynamics at arterial bifurcations in vivo. Neurosurgery. 59:1094–1100, 2006; discussion 1100–1101.
Meng, H., Z. Wang, Y. Hoi, L. Gao, E. Metaxa, D. D. Swartz, and J. Kolega. Complex hemodynamics at the apex of an arterial bifurcation induces vascular remodeling resembling cerebral aneurysm initiation. Stroke 38:1924–1931, 2007.
Meng, H., J. Xiang, and N. Liaw. The role of hemodynamics in intracranial aneurysm initiation. Int. Rev. Thrombosis. 7:40–57, 2012.
Metaxa, E., H. Meng, S. R. Kaluvala, M. P. Szymanski, R. A. Paluch, and J. Kolega. Nitric oxide-dependent stimulation of endothelial cell proliferation by sustained high flow. Am. J. Physiol. Heart Circ. Physiol. 295:H736–H742, 2008.
Metaxa, E., M. Tremmel, S. K. Natarajan, J. Xiang, R. A. Paluch, M. Mandelbaum, A. H. Siddiqui, J. Kolega, J. Mocco, and H. Meng. Characterization of critical hemodynamics contributing to aneurysmal remodeling at the basilar terminus in a rabbit model. Stroke 41:1774–1782, 2010.
Mintz, G. S., K. M. Kent, A. D. Pichard, L. F. Satler, J. J. Popma, and M. B. Leon. Contribution of inadequate arterial remodeling to the development of focal coronary artery stenoses. An intravascular ultrasound study. Circulation. 95:1791–1798, 1997.
Moore, Jr., J. E., E. Burki, A. Suciu, S. Zhao, M. Burnier, H. R. Brunner, and J. J. Meister. A device for subjecting vascular endothelial cells to both fluid shear stress and circumferential cyclic stretch. Ann. Biomed. Eng. 22:416–422, 1994.
Moriwaki, T., Y. Takagi, N. Sadamasa, T. Aoki, K. Nozaki, and N. Hashimoto. Impaired progression of cerebral aneurysms in interleukin-1beta-deficient mice. Stroke 37:900–905, 2006.
Murray, C. D. The physiological principle of minimum work: I. The vascular system and the cost of blood volume. Proc. Natl Acad. Sci. USA 12:207–214, 1926.
Nagata, I., H. Handa, N. Hashimoto, and F. Hazama. Experimentally induced cerebral aneurysms in rats: Part VI. Hypertension. Surg. Neurol. 14:477–479, 1980.
Nakatani, H., N. Hashimoto, Y. Kang, N. Yamazoe, H. Kikuchi, S. Yamaguchi, and H. Niimi. Cerebral blood flow patterns at major vessel bifurcations and aneurysms in rats. J. Neurosurg. 74:258–262, 1991.
Neelamegham, S., A. D. Taylor, A. R. Burns, C. W. Smith, and S. I. Simon. Hydrodynamic shear shows distinct roles for LFA-1 and Mac-1 in neutrophil adhesion to intercellular adhesion molecule-1. Blood 92:1626–1638, 1998.
Nuki, Y., M. M. Matsumoto, E. Tsang, W. L. Young, N. van Rooijen, C. Kurihara, and T. Hashimoto. Roles of macrophages in flow-induced outward vascular remodeling. J. Cereb. Blood Flow Metab. 29:495–503, 2009.
Park, J. B., F. Charbonneau, and E. L. Schiffrin. Correlation of endothelial function in large and small arteries in human essential hypertension. J. Hypertens. 19:415–420, 2001.
Rajagopalan, S., X. P. Meng, S. Ramasamy, D. G. Harrison, and Z. S. Galis. Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of vascular matrix metalloproteinases in vitro. Implications for atherosclerotic plaque stability. J. Clin. Invest. 98:2572–2579, 1996.
Rinkel, G. J., M. Djibuti, A. Algra, and J. van Gijn. Prevalence and risk of rupture of intracranial aneurysms: a systematic review. Stroke 29:251–256, 1998.
Ross, R. Atherosclerosis—an inflammatory disease. N. Engl. J. Med. 340:115–126, 1999.
Sakamoto, N., N. Saito, X. Han, T. Ohashi, and M. Sato. Effect of spatial gradient in fluid shear stress on morphological changes in endothelial cells in response to flow. Biochem. Biophys. Res. Commun. 395:264–269, 2010.
Samady, H., P. Eshtehardi, M. C. McDaniel, J. Suo, S. S. Dhawan, C. Maynard, L. H. Timmins, A. A. Quyyumi, and D. P. Giddens. Coronary artery wall shear stress is associated with progression and transformation of atherosclerotic plaque and arterial remodeling in patients with coronary artery disease. Circulation 124:779–788, 2011.
Schiffrin, E. L., J. B. Park, H. D. Intengan, and R. M. Touyz. Correction of arterial structure and endothelial dysfunction in human essential hypertension by the angiotensin receptor antagonist losartan. Circulation 101:1653–1659, 2000.
Schirmer, C. M., and A. M. Malek. Computational fluid dynamic characterization of carotid bifurcation stenosis in patient-based geometries. Brain Behav. 2:42–52, 2012.
Schirmer, C. M., and A. M. Malek. Wall shear stress gradient analysis within an idealized stenosis using non-Newtonian flow. Neurosurgery 61:853–863, 2007; discussion 863–864.
Schoenhagen, P., K. M. Ziada, S. R. Kapadia, T. D. Crowe, S. E. Nissen, and E. M. Tuzcu. Extent and direction of arterial remodeling in stable versus unstable coronary syndromes : an intravascular ultrasound study. Circulation 101:598–603, 2000.
Sho, E., M. Komatsu, M. Sho, H. Nanjo, T. M. Singh, C. Xu, H. Masuda, and C. K. Zarins. High flow drives vascular endothelial cell proliferation during flow-induced arterial remodeling associated with the expression of vascular endothelial growth factor. Exp. Mol. Pathol. 75:1–11, 2003.
Sho, E., M. Sho, T. M. Singh, H. Nanjo, M. Komatsu, C. Xu, H. Masuda, and C. K. Zarins. Arterial enlargement in response to high flow requires early expression of matrix metalloproteinases to degrade extracellular matrix. Exp. Mol. Pathol. 73:142–153, 2002.
Shumacker, Jr., H. B. Aneurysm development and degenerative changes in dilated artery proximal to arteriovenous fistula. Surg. Gynecol. Obstet. 130:636–640, 1970.
Singh, T. M., K. Y. Abe, T. Sasaki, Y. J. Zhuang, H. Masuda, and C. K. Zarins. Basic fibroblast growth factor expression precedes flow-induced arterial enlargement. J. Surg. Res. 77:165–173, 1998.
Slager, C. J., J. J. Wentzel, F. J. Gijsen, J. C. Schuurbiers, A. C. van der Wal, A. F. van der Steen, and P. W. Serruys. The role of shear stress in the generation of rupture-prone vulnerable plaques. Nat. Clin. Pract. Cardiovasc. Med. 2:401–407, 2005.
Stehbens, W. E. Aneurysms and anatomical variation of cerebral arteries. Arch. Pathol. 75:45–64, 1963.
Stone, P. H., A. U. Coskun, S. Kinlay, M. E. Clark, M. Sonka, A. Wahle, O. J. Ilegbusi, Y. Yeghiazarians, J. J. Popma, J. Orav, R. E. Kuntz, and C. L. Feldman. Effect of endothelial shear stress on the progression of coronary artery disease, vascular remodeling, and in-stent restenosis in humans: in vivo 6-month follow-up study. Circulation 108:438–444, 2003.
Szymanski, M. P., E. Metaxa, H. Meng, and J. Kolega. Endothelial cell layer subjected to impinging flow mimicking the apex of an arterial bifurcation. Ann. Biomed. Eng. 36:1681–1689, 2008.
Tada, Y., K. T. Kitazato, K. Yagi, K. Shimada, N. Matsushita, T. Kinouchi, Y. Kanematsu, J. Satomi, T. Kageji, and S. Nagahiro. Statins promote the growth of experimentally induced cerebral aneurysms in estrogen-deficient rats. Stroke 42:2286–2293, 2011.
Tada, Y., K. Yagi, K. T. Kitazato, T. Tamura, T. Kinouchi, K. Shimada, N. Matsushita, N. Nakajima, J. Satomi, T. Kageji, and S. Nagahiro. Reduction of endothelial tight junction proteins is related to cerebral aneurysm formation in rats. J. Hypertens. 28:1883–1891, 2010.
Tanweer, O., E. Metaxa, N. Liaw, S. Sternberg, A. Siddiqui, J. Kolega, and H. Meng. Inhibition of stretch-activated ion channels on endothelial cells disrupts nitric oxide-mediated arterial outward remodeling. J. Biorheol. 24:77–83, 2011.
Teng, Z., G. Canton, C. Yuan, M. Ferguson, C. Yang, X. Huang, J. Zheng, P. K. Woodard, and D. Tang. 3D critical plaque wall stress is a better predictor of carotid plaque rupture sites than flow shear stress: an in vivo MRI-based 3D FSI study. J. Biomech. Eng. 132:031007, 2010.
Thi, M. M., J. M. Tarbell, S. Weinbaum, and D. C. Spray. The role of the glycocalyx in reorganization of the actin cytoskeleton under fluid shear stress: a “bumper-car” model. Proc. Natl Acad. Sci. USA 101:16483–16488, 2004.
Tohda, K., H. Masuda, K. Kawamura, and T. Shozawa. Difference in dilatation between endothelium-preserved and -desquamated segments in the flow-loaded rat common carotid artery. Arterioscler. Thromb. 12:519–528, 1992.
Torii, R., N. B. Wood, N. Hadjiloizou, A. W. Dowsey, A. R. Wright, A. D. Hughes, J. Davies, D. P. Francis, J. Mayet, G. Z. Yang, S. A. Thom, and X. Y. Xu. Stress phase angle depicts differences in coronary artery hemodynamics due to changes in flow and geometry after percutaneous coronary intervention. Am. J. Physiol. Heart Circ. Physiol. 296:H765–H776, 2009.
Tremmel, M., J. Xiang, S. K. Natarajan, L. N. Hopkins, A. H. Siddiqui, E. I. Levy, and H. Meng. Alteration of intra-aneurysmal hemodynamics for flow diversion using enterprise and vision stents. World Neurosurg. 74:306–315, 2010.
Tronc, F., Z. Mallat, S. Lehoux, M. Wassef, B. Esposito, and A. Tedgui. Role of matrix metalloproteinases in blood flow-induced arterial enlargement: interaction with NO. Arterioscler. Thromb. Vasc. Biol. 20:E120–E126, 2000.
Tronc, F., M. Wassef, B. Esposito, D. Henrion, S. Glagov, and A. Tedgui. Role of NO in flow-induced remodeling of the rabbit common carotid artery. Arterioscler. Thromb. Vasc. Biol. 16:1256–1262, 1996.
Tuttle, J. L., R. D. Nachreiner, A. S. Bhuller, K. W. Condict, B. A. Connors, B. P. Herring, M. C. Dalsing, and J. L. Unthank. Shear level influences resistance artery remodeling: wall dimensions, cell density, and eNOS expression. Am. J. Physiol. Heart Circ. Physiol. 281:H1380–H1389, 2001.
Tzima, E., M. Irani-Tehrani, W. B. Kiosses, E. Dejana, D. A. Schultz, B. Engelhardt, G. Cao, H. DeLisser, and M. A. Schwartz. A mechanosensory complex that mediates the endothelial cell response to fluid shear stress. Nature 437:426–431, 2005.
van Everdingen, K. J., C. J. Klijn, L. J. Kappelle, W. P. Mali, and J. van der Grond. MRA flow quantification in patients with a symptomatic internal carotid artery occlusion. The Dutch EC-IC Bypass Study Group. Stroke 28:1595–1600, 1997.
Van Remmen, H., M. D. Williams, Z. Guo, L. Estlack, H. Yang, E. J. Carlson, C. J. Epstein, T. T. Huang, and A. Richardson. Knockout mice heterozygous for Sod2 show alterations in cardiac mitochondrial function and apoptosis. Am. J. Physiol. Heart Circ. Physiol. 281:H1422–H1432, 2001.
Varnava, A. M., P. G. Mills, and M. J. Davies. Relationship between coronary artery remodeling and plaque vulnerability. Circulation 105:939–943, 2002.
Vega, C., J. V. Kwoon, and S. D. Lavine. Intracranial aneurysms: current evidence and clinical practice. Am. Fam. Physician 66:601–608, 2002.
Waga, S., M. Okada, and T. Kojima. Saccular aneurysm associated with absence of the left cervical carotid arteries. Neurosurgery. 3:208–212, 1978.
Wang, Z., J. Kolega, Y. Hoi, L. Gao, D. D. Swartz, E. I. Levy, J. Mocco, and H. Meng. Molecular alterations associated with aneurysmal remodeling are localized in the high hemodynamic stress region of a created carotid bifurcation. Neurosurgery 65:169–177, 2009; discussion 177–178.
Weinberg, P. D., and C. Ross Ethier. Twenty-fold difference in hemodynamic wall shear stress between murine and human aortas. J. Biomech. 40:1594–1598, 2007.
Wentzel, J. J., Y. S. Chatzizisis, F. J. Gijsen, G. D. Giannoglou, C. L. Feldman, and P. H. Stone. Endothelial shear stress in the evolution of coronary atherosclerotic plaque and vascular remodeling: current understanding and remaining questions. Cardiovasc. Res. 96(2):234–243, 2012.
White, S. J., E. M. Hayes, S. Lehoux, J. Y. Jeremy, A. J. Horrevoets, and A. C. Newby. Characterization of the differential response of endothelial cells exposed to normal and elevated laminar shear stress. J. Cell. Physiol. 226:2841–2848, 2011.
Yong-Zhong, G., and H. A. van Alphen. Pathogenesis and histopathology of saccular aneurysms: review of the literature. Neurol. Res. 12:249–255, 1990.
Zarins, C. K., D. P. Giddens, B. K. Bharadvaj, V. S. Sottiurai, R. F. Mabon, and S. Glagov. Carotid bifurcation atherosclerosis. Quantitative correlation of plaque localization with flow velocity profiles and wall shear stress. Circ. Res. 53:502–514, 1983.
Zarins, C. K., M. A. Zatina, D. P. Giddens, D. N. Ku, and S. Glagov. Shear stress regulation of artery lumen diameter in experimental atherogenesis. J. Vasc. Surg. 5:413–420, 1987.
Zhao, S., A. Suciu, T. Ziegler, J. E. Moore, Jr., E. Burki, J. J. Meister, and H. R. Brunner. Synergistic effects of fluid shear stress and cyclic circumferential stretch on vascular endothelial cell morphology and cytoskeleton. Arterioscler. Thromb. Vasc. Biol. 15:1781–1786, 1995.
Acknowledgments
We thank Nicholas Liaw for critical review of the manuscript and assistance with figures, and Chris Martensen for assistance with figures and references. This work was supported by NIH grant R01NS064592 (awarded to H.M.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Seungik Baek oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Dolan, J.M., Kolega, J. & Meng, H. High Wall Shear Stress and Spatial Gradients in Vascular Pathology: A Review. Ann Biomed Eng 41, 1411–1427 (2013). https://doi.org/10.1007/s10439-012-0695-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-012-0695-0