Abstract
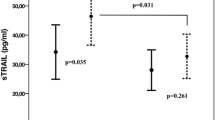
Different postconditioning (Postcon) methods have been demonstrated to protect heart from ischemia/reperfusion injury. The relationship between Postcon by percutaneous coronary intervention (PCI) and apoptosis is not clear. Our objective was to test whether Postcon by PCI in patients with acute myocardial infarction (AMI) reduces myocardial apoptosis. Seventy-five patients were randomly assigned to one of three groups before stenting. The Routine group (n = 26) received no Postcon intervention prior to the onset of reperfusion; Postcon-30s (n = 25) and Postcon-60s groups (n = 24) underwent three cycles of 30- or 60-s balloon deflation and 30- or 60-s inflation. Additionally, 34 normal controls (NC) were enrolled in the study. Plasma concentrations of soluble Fas/APO-1 ([sFas]) and Fas ligand ([sFasL]) were determined at baseline and 7 days after PCI via ELISA. The [sFas] and [sFasL] in AMI patients were significantly elevated at baseline as compared with NC (P < 0.01), and showed an upward trend in the Routine group, a slightly upward trend in Postcon-30s, and a downward trend in Postcon-60s at 7 days. Comparison among the three groups showed significant differences (P < 0.05, 3.8 vs. 4.6 vs. 5.1 ng ml−1). The [sFasL] in Postcon-60s was significantly decreased at 7 days (P < 0.05, 3.9 vs. 3.1 ng ml−1) compared with baseline, but not Postcon-30s and Routine. More importantly, Postcon-60s group had the lowest [sFasL], followed by Postcon-30s, which had a lower value than Routine at 7 days (P < 0.05, 3.1 vs.3.7 vs. 4.2 ng ml−1). Our results suggest that Postcon-60s was visibly better than Postcon-30s, which in turn was better than Routine for inhibition of the effects of myocardial apoptosis and reduction of reperfusion injury in patients with acute myocardial infarction.



Similar content being viewed by others
References
Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, Vinten-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285:H579–H588
Kin H, Zhao ZQ, Sun HY, Wang NP, Corvera JS, Halkos ME, Kerendi F, Guyton RA, Vinten-Johansen J (2004) Postconditioning attenuates myocardial ischemia-reperfusion injury by inhibiting events in the early minutes of reperfusion. Cardiovasc Res 62:74–85
Argaud L, Gateau-Roesch O, Raisky O, Loufouat J, Robert D, Ovize M (2005) Postconditioning inhibits mitochondrial permeability transition. Circulation 111:194–197
Andreka G, Vertesaljai M, Szantho G, Font G, Piroth Z, Fontos G, Juhasz ED, Szekely L, Szelid Z, Turner MS, Ashrafian H, Frenneaux MP, Andreka P (2007) Remote ischaemic postconditioning protects the heart during acute myocardial infarction in pigs. Heart 93:749–752
Baxter GF, Yellon DM (2003) Current trends and controversies in ischemia-reperfusion research—meeting report of the Hatter institute 3rd international workshop on cardioprotection. Basic Res Cardiol 98:133–136
Staat P, Rioufol G, Piot C, Cottin Y, Cung TT, L’Huillier I, Aupetit JF, Bonnefoy E, Finet G, ndre-Fouet X, Ovize M (2005) Postconditioning the human heart. Circulation 112:2143–2148
Ma XJ, Zhang XH, Li CM, Luo M (2006) Effect of postconditioning on coronary blood flow velocity and endothelial function in patients with acute myocardial infarction. Scand Cardiovasc J 40:327–333
Dow J, Kloner RA (2007) Postconditioning does not reduce myocardial infarct size in an in vivo regional ischemia rodent model. J Cardiovasc Pharmacol Ther 12:153–163
Manintveld OC, Lintel Te, Hekkert M, van den Bos EJ, Suurenbroek GM, Dekkers DH, Verdouw PD, Lamers JM, Duncker DJ (2007) Cardiac effects of postconditioning depend critically on the duration of index ischemia. Am J Physiol Heart Circ Physiol 292:H1551–H1560
Yang XC, Liu Y, Wang LF, Cui L, Wang T, Ge YG, Wang HS, Li WM, Xu L, Ni ZH, Liu SH, Zhang L, Jia HM, Vinten-Johansen J, Zhao ZQ (2007) Reduction in myocardial infarct size by postconditioning in patients after percutaneous coronary intervention. J Invasive Cardiol 19:424–430
Vinten-Johansen J, Zhao ZQ, Zatta AJ, Kin H, Halkos ME, Kerendi F (2005) Postconditioning—a new link in nature’s armor against myocardial ischemia-reperfusion injury. Basic Res Cardiol 100:295–310
Zhang L, Zhang L, Li YH, Zhang HY, Chen ML, Gao MM, Hu AH, Yang HS, Yang HS (2005) High-dose glucose-insulin-potassium treatment reduces myocardial apoptosis in patients with acute myocardial infarction. Eur J Clin Invest 35:164–170
Shimizu M, Fukuo K, Nagata S, Suhara T, Okuro M, Fujii K, Higashino Y, Mogi M, Hatanaka Y, Ogihara T (2002) Increased plasma levels of the soluble form of fas ligand in patients with acute myocardial infarction and unstable angina pectoris. J Am Coll Cardiol 39:585–590
Jelachich ML, Lipton HL (2001) Theiler’s murine encephalomyelitis virus induces apoptosis in gamma interferon-activated M1 differentiated myelomonocytic cells through a mechanism involving tumor necrosis factor alpha (TNF-alpha) and TNF-alpha-related apoptosis-inducing ligand. J Virol 75:5930–5938
Suda T, Takahashi T, Golstein P, Nagata S (1993) Molecular cloning and expression of the fas ligand, a novel member of the tumor necrosis factor family. Cell 75:1169–1178
Cicala S, de Simone G, Roman MJ, Best LG, Lee ET, Wang W, Welty TK, Galloway JM, Howard BV, Devereux RB (2007) Prevalence and prognostic significance of wall-motion abnormalities in adults without clinically recognized cardiovascular disease: the strong heart study. Circulation 116:143–150
Piper HM, Abdallah Y, Schafer C (2004) The first minutes of reperfusion: a window of opportunity for cardioprotection. Cardiovasc Res 61:365–371
Heusch G (2004) Postconditioning: old wine in a new bottle? J Am Coll Cardiol 44:1111–1112
Searle J, Kerr JF, Bishop CJ (1982) Necrosis and apoptosis: distinct modes of cell death with fundamentally different significance. Pathol Annu 17(Pt 2):229–259
Saraste A, Pulkki K, Kallajoki M, Henriksen K, Parvinen M, Voipio-Pulkki LM (1997) Apoptosis in human acute myocardial infarction. Circulation 95:320–323
Cheng W, Kajstura J, Nitahara JA, Li B, Reiss K, Liu Y, Clark WA, Krajewski S, Reed JC, Olivetti G, Anversa P (1996) Programmed myocyte cell death affects the viable myocardium after infarction in rats. Exp Cell Res 226:316–327
Veinot JP, Gattinger DA, Fliss H (1997) Early apoptosis in human myocardial infarcts. Hum Pathol 28:485–492
Tanaka M, Ito H, Adachi S, Akimoto H, Nishikawa T, Kasajima T, Marumo F, Hiroe M (1994) Hypoxia induces apoptosis with enhanced expression of fas antigen messenger RNA in cultured neonatal rat cardiomyocytes. Circ Res 75:426–433
Wang HC, Zhang HF, Guo WY, Su H, Zhang KR, Li QX, Yan W, Ma XL, Lopez BL, Christopher TA, Gao F (2006) Hypoxic postconditioning enhances the survival and inhibits apoptosis of cardiomyocytes following reoxygenation: role of peroxynitrite formation. Apoptosis 11:1453–1460
Sun HY, Wang NP, Halkos M, Kerendi F, Kin H, Guyton RA, Vinten-Johansen J, Zhao ZQ (2006) Postconditioning attenuates cardiomyocyte apoptosis via inhibition of JNK and p38 mitogen-activated protein kinase signaling pathways. Apoptosis 11:1583–1593
Vinten-Johansen J (2007) Postconditioning: a mechanical maneuver that triggers biological and molecular cardioprotective responses to reperfusion. Heart Fail Rev 12:235–244
Thibault H, Piot C, Staat P, Bontemps L, Sportouch C, Rioufol G, Cung TT, Bonnefoy E, Angoulvant D, Aupetit JF, Finet G, ndre-Fouet X, Macia JC, Raczka F, Rossi R, Itti R, Kirkorian G, Derumeaux G, Ovize M (2008) Long-term benefit of postconditioning. Circulation 117:1037–1044
Griffiths EJ, Halestrap AP (1995) Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J 307:93–98
Gomez L, Paillard M, Thibault H, Derumeaux G, Ovize M (2008) Inhibition of GSK3beta by postconditioning is required to prevent opening of the mitochondrial permeability transition pore during reperfusion. Circulation 117:2761–2768
Schwartz LM, Lagranha CJ (2006) Ischemic postconditioning during reperfusion activates Akt and ERK without protecting against lethal myocardial ischemia-reperfusion injury in pigs. Am J Physiol Heart Circ Physiol 290:H1011–H1018
Iliodromitis EK, Georgiadis M, Cohen MV, Downey JM, Bofilis E, Kremastinos DT (2006) Protection from post-conditioning depends on the number of short ischemic insults in anesthetized pigs. Basic Res Cardiol 101:502–507
Shliakhto EV, Galagudza MM, Syrenskii AV, Tsyrlin VA (2005) Ischemic postconditioning of the myocardium: a new method of heart protection against reperfusion damage. Ter Arkh 77:77–80
Mykytenko J, Kerendi F, Reeves JG, Kin H, Zatta AJ, Jiang R, Guyton RA, Vinten-Johansen J, Zhao ZQ (2007) Long-term inhibition of myocardial infarction by postconditioning during reperfusion. Basic Res Cardiol 102:90–100
Author information
Authors and Affiliations
Corresponding authors
Additional information
Wen-Shu Zhao and Lin Xu contributed equally to the investigation.
Rights and permissions
About this article
Cite this article
Zhao, WS., Xu, L., Wang, LF. et al. A 60-s postconditioning protocol by percutaneous coronary intervention inhibits myocardial apoptosis in patients with acute myocardial infarction. Apoptosis 14, 1204–1211 (2009). https://doi.org/10.1007/s10495-009-0387-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-009-0387-x