Abstract
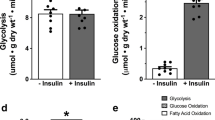
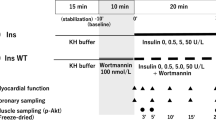
This paper summarizes data from different studies all aimed at elucidating regulation of protein kinase B in the diabetic heart. Two rat models of type 2 diabetes mellitus ((i) elicited via neonatal streptozotocin injection (Stz) and (ii) Zucker fa/fa rats), were used as well as different experimental models viz isolated, Langendorff perfused hearts as well as adult ventricular myocytes. Glucose uptake was elicited by a variety of stimuli and the activation of PKB measured in tandem. Basal glucose uptake was impaired in both diabetes models while basal phosphorylation of PKB differed, showing lower levels in the Stz model but higher levels in the Zucker rats. Neither 100 nM insulin nor 10–8 M isoproterenol could stimulate PKB phosphorylation to the same extent in the diabetic myocardium as in controls, regardless of the method used, but a combination of these stimuli resulted in an additive response. Concurrent glucose uptake however, was not additive. Wortmannin abolished both insulin and isoproterenol stimulation of glucose uptake as well as PKB phosphorylation. In contrast to the above-mentioned results, the protein tyrosine phosphatase inhibitor vanadate, alone or in combination with insulin, elicited PKB phosphorylation to the same extent in diabetic cardiomyocytes as in controls. Despite this, glucose uptake stimulated by vanadate or insulin in combination with vanadate was attenuated. The combination of insulin and vanadate may however be beneficial to the diabetic heart as it resulted in improved glucose transport. Results from the different studies can be summarized as follows: (i) dysregulation of PKB is evident in the diabetic myocardium, (ii) PKB activation is not always directly correlated with glucose uptake and (iii) insulin resistance is associated with multiple alterations in signal transduction, both above and below PKB activation.
Similar content being viewed by others
References
Tanti JF, Grillo S, Gremeaux T, Coffer PJ, Obberghen EV, Marchand-Brustel YL: Potential role of protein kinase B in glucose transporter 4 translocation in adipocytes. Endocrinology 138: 2005-2010, 1997
Kohn AD, Summers SA, Birnbaum MJ, Roth RA: Expression of a constitutively active Akt Ser/Thr kinase in 3T3-L1 adipocytes stimulates glucose uptake and glucose transporter 4 translocation. J Biol Chem 271: 31372-31378, 1996
Ueki K, Yamamoto-Honda R, Kaburagi Y et al.: Potential role of protein kinase B in insulin-induced glucose transport, glycogen synthesis, and protein synthesis. J Biol Chem 273: 5315-5322, 1998
Hajduch E, Alessi DR, Hemmings BA, Hundal HS: Constitutive activation of protein kinase B alpha by membrane targeting promotes glucose and system A amino acid transport, protein synthesis, and activation of glycogen synthase kinase 3 in L6 muscle cells. Diabetes 47: 1006-1013, 1998
Han X, Bonen A: Epinephrine translocates GLUT-4 but inhibits insulin-stimulated glucose transport in rat muscle. Am J Physiol 274: E700-E704, 1998
Doenst T, Taegtmeyer H: Alpha-adrenergic stimulation mediates glucose uptake through phosphatidylinositol 3-kinase in rat heart. Circ Res 84: 467-474, 1999
Shimizu Y, Kielar D, Minokoshi Y, Shimazu T: Noradrenaline increases glucose transport into brown adipocytes in culture by a mechanism different from that of insulin. Biochem J 314: 485-490, 1996
Kohn AD, Kovacina K, Roth RA: Insulin stimulates the kinase activity of RAC-PK, a pleckstrin homology domain containing Ser/Thr kinase. EMBO J 14: 4288-4295, 1995
Shimizu Y, Kielar D, Minokoshi Y, Shimazu T: Noradrenaline increases glucose transport into brown adipocytes in culture by a mechanism different from that of insulin. Biochem J 314: 485-490, 1996
Sable CL, Filippa N, Hemmings B, Van Obberghen E: cAMP stimulates protein kinase B in a Wortmannin-insensitive manner. FEBS Lett 409: 253-257, 1997
Morisco C, Zebrowski D, Condorelli G, Tsichlis P, Vatner SF, Sadoshima J: The Akt-glycogen synthase kinase 3β pathway regulates transcription of atrial natriuretic factor induced by β-adrenergic receptor stimulation in cardiac myocytes. J Biol Chem 275: 14466-14475, 2000
Kopp SJ, Daar J, Paulson DJ, Ramano FD, Laddaga R: Effects of oral vanadyl treatment on diabetes-induced alterations in the heart GLUT-4 transporter. J Mol Cell Cardiol 29: 2355-2362, 1997
Donthi RV, Huisamen B, Lochner A: Effect of vanadate and insulin on glucose transport in isolated adult rat cardiomyocytes. Cardiovasc Drugs Therapy 14: 463-470, 2000
Portha B, Blondel O, Serradas P, McEvoy R, Giroix M-H, Kergoat M, Bailbe D: The rat models of non-insulin dependent diabetes induced by neonatal streptozotocin. Diabetes and Metabolism 15: 61-75, 1989
Fischer Y, Rose H, Kammermeir H: Highly insulin-responsive isolated rat heart muscle cells yielded by a modified isolation method. Life Sci 49: 1679-1688, 1991
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 193: 265-275, 1951
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 71: 248-254, 1976
Zaninetti D, Crettax M, Jeanrenaud B: Dysregulation of glucose transport in hearts of genetically obese (fa/fa) rats. Diabetologia 25: 525-529, 1983
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA: Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 15: 6541-6551, 1996
Huisamen B, Van Dyk M, Keyser A, Lochner A: The effects of insulin and β-adrenergic stimulation on glucose transport, glut 4 and PKB activation in the myocardium of lean and obese non-insulin dependent diabetes mellitus rats. J Mol Cell Biochem 223: 15-25, 2001
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huisamen, B. Protein kinase B in the diabetic heart. Mol Cell Biochem 249, 31–38 (2003). https://doi.org/10.1023/A:1024749614222
Issue Date:
DOI: https://doi.org/10.1023/A:1024749614222