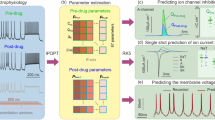
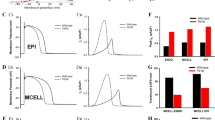
Abstract
Advances in genetics and molecular biology have provided an extensive body of information on the structure and function of the elementary building blocks of living systems. Genetic defects in membrane ion channels can disrupt the delicate balance of dynamic interactions between the ion channels and the cellular environment, leading to altered cell function1,2,3. As ion-channel defects are typically studied in isolated expression systems, away from the cellular environment where they function physiologically, a connection between molecular findings and the physiology and pathophysiology of the cell is rarely established. Here we describe a single-channel-based Markovian modelling approach that bridges this gap. We achieve this by determining the cellular arrhythmogenic consequences of a mutation in the cardiac sodium channel that can lead to a clinical arrhythmogenic disorder (the long-QT syndrome) and sudden cardiac death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others

References
Welsh, M. J. & Hoshi, T. Ion channels lose the rhythm. Nature 376, 640 (1995).
Attali, B. Anew wave for heart rhythms. Nature 384, 24–25 (1996).
Keating, M. T. & Sanguinetti, M. C. Molecular genetic insights into cardiovascular disease. Science 272, 681–685 (1996).
Barinaga, M. Tracking down mutations that can stop the heart. Science 281, 32–34 (1998).
Dumaine, R. et al. Multiple-mechanisms of Na+ channel-linked long-QT syndrome. Circ. Res. 78, 916–924 (1996).
Chandra, R., Starmer, C. F. & Grant, A. O. Multiple effects of the KPQ deletion on gating of human cardiac Na+ channels expressed in mammalian cells. Am. J. Physiol. 274, H1643–H1654 (1998).
Bennett, P. B., Yazawa, K., Makita, N. & George, A. L. J Molecular mechanism for an inherited cardiac arrhythmia. Nature 376, 683–685 (1995).
Luo, C. H. & Rudy, Y. Adynamic model of the cardiac ventricular action potential. I. Simulations of ionic currents and concentration changes. Circ. Res. 74, 1071–1096 (1994).
Luo, C. H. & Rudy, Y. Adynamic model of the cardiac ventricular action potential. II. Afterdepolarizations, triggered activity, and potentiation. Circ. Res. 74, 1097–1113 (1994).
Zeng, J., Laurita, K. R., Rosenbaum, D. S. & Rudy, Y. Two components of the delayed rectifier K+ current in ventricular myocytes of the guinea pig type. Theoretical formulation and their role in repolarization. Circ. Res. 77, 140–152 (1995).
Wamg. D. W. Yazawa, K., George, A. L. J & Bennett, P. B. Characterization of human cardiac Na+ channel mutations in the congenital long QT syndrome. Proc. Natl Acad. Sci. USA 93, 13200–13205 (1996).
Yue, D. T., Lawrence, J. H. & Marban, E. Two molecular transitions influence cardiac sodium channel gating. Science 244, 349–352 (1989).
Wang, D. W., Yazawa, K., Makita, N., George, A. L. J & Bennett, P. B. Pharmacological targeting of long QT mutant sodium channels. J. Clin. Invest. 99, 1714–1720 (1997).
Zeng, J. & Rudy, Y. Early afterdepolarizations in cardiac myocytes: mechanism and rate dependence. Biophys. J. 68, 949–964 (1995).
Schwartz, P. J. et al. Long QT syndrome patients with mutations of the SCN5A and HERG genes have differential responses to Na+ channel blockade and to increases in heart rate. Implications for gene-specific therapy. Circulation 92, 3381–3386 (1995).
Hodgkin, A. L. & Huxley, A. F. Aquantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 117, 500–544 (1952).
Tomaselli, G. F. et al. Amutation in the pore of the sodium channel alters gating. Biophys. J. 68, 1814–1827 (1995).
Mitsuiye, T. & Noma, A. Inactivation of the cardiac Na+ channels in guinea-pig ventricular cells through the open state. J. Physiol. 485, 581–594 (1995); erratum J. Physiol. (Lond.) 488, 841 (1995).
Hanck, D. A. in Cardiac Electrophysiology: From Cell to Bedside(eds Zipes, W. & Jalife, J.) 65–74 (W. B. Saunders, Philadelphia, (1995).
Balser, J. R., Roden, D. M. & Bennett, P. B. Global parameter optimization for cardiac potassium channel gating models. Biophys. J. 57, 433–444 (1990).
Zeng, J. in Biomedical Engineering(Case Western Reserve Univ., Cleveland, (1997).
Acknowledgements
We thank P. Bennett and A. Grant for providing us with original figures from their experimental publications. This work was supported by the NIH-National Heart, Lung and Blood Institute and The Whitaker Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Clancy, C., Rudy, Y. Linking a genetic defect to its cellular phenotype in a cardiac arrhythmia. Nature 400, 566–569 (1999). https://doi.org/10.1038/23034
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/23034
This article is cited by
-
Certain beta blockers (e.g., bisoprolol) may be reevaluated in hypertension guidelines for patients with left ventricular hypertrophy to diminish the ventricular arrhythmic risk
Journal of Human Hypertension (2021)
-
Autonomic modulation of ventricular electrical activity: recent developments and clinical implications
Clinical Autonomic Research (2021)
-
Computational models in cardiology
Nature Reviews Cardiology (2019)
-
Combination of “quadratic adaptive algorithm” and “hybrid operator splitting” or uniformization algorithms for stability against acceleration in the Markov model of sodium ion channels in the ventricular cell model
Medical & Biological Engineering & Computing (2019)
-
A Mathematical Model of the Human Cardiac Na+ Channel
The Journal of Membrane Biology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.