Abstract
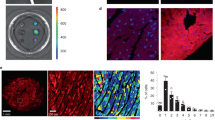
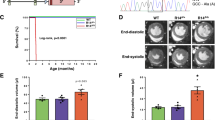
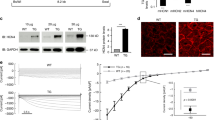
The inwardly rectifying K+ channel Kir6.1 forms K+ channels by coupling with a sulfonylurea receptor in reconstituted systems, but the physiological roles of Kir6.1-containing K+ channels have not been determined. We report here that mice lacking the gene encoding Kir6.1 (known as Kcnj8) have a high rate of sudden death associated with spontaneous ST elevation followed by atrioventricular block as seen on an electrocardiogram. The K+ channel opener pinacidil did not induce K+ currents in vascular smooth-muscle cells of Kir6.1-null mice, and there was no vasodilation response to pinacidil. The administration of methylergometrine, a vasoconstrictive agent, elicited ST elevation followed by cardiac death in Kir6.1-null mice but not in wild-type mice, indicating a phenotype characterized by hypercontractility of coronary arteries and resembling Prinzmetal (or variant) angina in humans. The Kir6.1-containing K+ channel is critical in the regulation of vascular tonus, especially in the coronary arteries, and its disruption may cause Prinzmetal angina.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jan, L.Y. & Jan, Y.N. Voltage-gated and inwardly rectifying potassium channels. J. Physiol. 505, 267–282 (1997).
Nichols, C.G. & Lopatin, A.N. Inward rectifier potassium channels. Annu. Rev. Physiol. 59, 171–191 (1997).
Abraham, M.R., Jahangir, A., Alekseev, A.E. & Terzic, A. Channelopathies of inwardly rectifying potassium channels. FASEB J. 13, 1901–1910 (1999).
Inagaki, N. et al. Cloning and functional characterization of a novel ATP-sensitive potassium channel ubiquitously expressed in rat tissues, including pancreatic islets, pituitary, skeletal muscle, and heart. J. Biol. Chem. 270, 5691–5694 (1995).
Inagaki, N. et al. Reconstitution of IKATP: An inward rectifier subunit plus the sulfonylurea receptor. Science 270, 1166–1170 (1995).
Ashcroft, F.M. & Gribble, F.M. Correlating structure and function in ATP-sensitive K+ channels. Trends. Neurosci. 21, 288–294 (1998).
Aguilar-Bryan, L. & Bryan, J. Molecular biology of adenosine triphosphate-sensitive potassium channels. Endocr. Rev. 20, 101–135 (1999).
Seino, S. ATP-sensitive potassium channels: A model of heteromultimeric potassium channel/receptor assemblies. Annu. Rev. Physiol. 61, 337–362 (1999).
Inagaki, N. et al. A family of sulfonylurea receptors determines the pharmacological properties of ATP-sensitive K+ channels. Neuron 16, 1011–1017 (1996).
Isomoto, S. et al. A novel sulfonylurea receptor forms with BIR (Kir6.2) a smooth muscle type ATP-sensitive K+ channel. J. Biol. Chem. 271, 24321–24324 (1996).
Miki, T. et al. Defective insulin secretion and enhanced insulin action in KATP channel-deficient mice. Proc. Natl. Acad. Sci. USA. 95, 10402–10406 (1998).
Li, R.A., Leppo, M., Miki, T., Seino, S. & Marban, E. Molecular basis of electrocardiographic ST-segment elevation. Circ. Res. 87, 837–839 (2000).
Suzuki, M. et al. Functional roles of cardiac and vascular ATP-sensitive potassium channels clarified by Kir6.2-knockout mice. Circ. Res. 88, 570–577 (2001).
Miki, T. et al. ATP-sensitive K+ channels in the hypothalamus are essential for the maintenance of glucose homeostasis. Nature Neurosci. 4, 507–512 (2001).
Suzuki, M. et al. Role of sarcolemmal KATP channels in cardioprotection against ischemia/reperfusion injury in mice. J. Clin. Invest. 109, 509–516 (2002).
Ämmälä, C., Moorhouse, A. & Ashcroft, F.M. The sulphonylurea receptor confers diazoxide sensitivity on the inwardly rectifying K+ channel Kir6.1 expressed in human embryonic kidney cells. J. Physiol. 494, 709–714 (1996).
Kono, Y. et al. The properties of the Kir6.1-6.2 tandem channel co-expressed with SUR2A. Pflugers. Arch. 440, 692–698 (2000).
Yamada, M. et al. Sulphonylurea receptor 2B and Kir6.1 form a sulphonylurea-sensitive but ATP-insensitive K+ channel. J. Physiol. 499, 715–720 (1997).
Standen, N.B. et al. Hyperpolarizing vasodilators activate ATP-sensitive K+ channels in arterial smooth muscle. Science 245, 177–180 (1989).
Kajioka, S., Kitamura, K. & Kuriyama, H. Guanosine diphosphate activates an adenosine 5′-triphosphate-sensitive K+ channel in the rabbit portal vein. J. Physiol. 444, 397–418 (1991).
Beech, D.J., Zhang, H., Nakao, K. & Bolton, T.B. K channel activation by nucleotide diphosphates and its inhibition by glibenclamide in vascular smooth-muscle cells. Br. J. Pharmacol. 110, 573–582 (1993).
Prinzmetal, M. et al. Angina pectoris. 1. A variant form of angina pectoris: Preliminary report. Am. J. Med. 27, 375–378 (1959).
Maseri, A. Role of coronary artery spasm in symptomatic and silent myocardial ischemia. J. Am. Coll. Cardiol. 9, 249–262 (1987).
Liu, Y., Sato, T., O'Rourke, B. and Marban, E. Mitochondrial ATP-dependent potassium channels: Novel effectors of cardioprotection? Circulation 97, 2463–2469 (1998).
Egashira, K. et al. Mechanism of ergonovine-induced hyperconstriction of the large epicardial coronary artery in conscious dogs a month after arterial injury. Circ. Res. 71, 435–442 (1992).
Myerburg, R.J., Interian, A., Jr Mitrani, R.M., Kessler, K.M. & Castellanos, A. Frequency of sudden cardiac death and profiles of risk. Am. J. Cardiol. 80, 10F–19F (1997).
Keating, M.T. & Sanguinetti, M.C. Molecular and cellular mechanisms of cardiac arrhythmias. Cell 104, 569–580 (2001).
Gutstein, D.E. et al. Conduction slowing and sudden arrhythmic death in mice with cardiac-restricted inactivation of connexin43. Circ. Res. 88, 333–339 (2001).
Robbins, J. & Dorn, G.W. 2nd. Listening for hoof beats in heart beats. Nature Med. 6, 968–970 (2000).
Kupershmidt, S. et al. Replacement by homologous recombination of the minK gene with lacZ reveals restriction of minK expression to the mouse cardiac conduction system. Circ. Res. 84, 146–152 (1999).
Drici, M.D. et al. Involvement of IsK-associated K+ channel in heart rate control of repolarization in a murine engineered model of Jervell and Lange-Nielsen syndrome. Circ. Res. 83, 95–102 (1998).
Barry, D.M., Xu, H., Schuessler, R.B. & Nerbonne, J.M. Functional knockout of the transient outward current, long-QT syndrome, and cardiac remodeling in mice expressing a dominant-negative Kv4 α subunit. Circ. Res. 83, 560–567 (1998).
Seharaseyon, J., et al. Evidence against functional heteromultimerization of the KATP channel subunits Kir6.1 and Kir6.2. J. Biol. Chem. 275, 17561–17565 (2000).
Suzuki, M. et al. Kir6.1: A possible subunit of ATP-sensitive K+ channels in mitochondria. Biochem. Biophys. Res. Commun. 241, 693–697 (1997).
Thomzig, A. et al. Kir6.1 is the principal pore-forming subunit of astrocyte but not neuronal plasma membrane K-ATP channels. Mol. Cell. Neurosci. 18, 671–690 (2001).
Okumura, K. et al. Diffuse disorder of coronary artery vasomotility in patients with coronary spastic angina. Hyperreactivity to the constrictor effects of acetylcholine and the dilator effects of nitroglycerin. J. Am. Coll. Cardiol. 27, 45–52 (1996).
MacAlpin, R.N. Cardiac arrest and sudden unexpected death in variant angina: Complications of coronary spasm that can occur in the absence of severe organic coronary stenosis. Am. Heart J. 125, 1011–1017 (1993).
Beltrame, J.F., Sasayama, S. & Maseri, A. Racial heterogeneity in coronary artery vasomotor reactivity: Differences between Japanese and Caucasian patients. J. Am. Coll. Cardiol. 33, 1442–1452 (1999).
Pristipino, C. et al. Major racial differences in coronary constrictor response between Japanese and Caucasians with recent myocardial infarction. Circulation 101, 1102–1108 (2000).
Tanaka, J. et al. Cellular distribution of the P2X4 ATP receptor mRNA in the brain and non-neuronal organs of rats. Arch. Histol. Cytol. 59, 485–490 (1996).
Acknowledgements
We thank M. Tamagawa and Y. Reien for technical assistance. This work was supported by Grants-in-Aid for Creative Scientific Research (10NP0201) and for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology; by a Scientific Research Grant from the Ministry of Health, Labour and Welfare, Japan; and by grants from the Uehara Memorial Foundation, from the Kanae Foundation, from Mitsui Life Insurance Research Foundation, from K. Watanabe Research Fund, and from the Yamanouchi Foundation for Research on Metabolic Disorders.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Miki, T., Suzuki, M., Shibasaki, T. et al. Mouse model of Prinzmetal angina by disruption of the inward rectifier Kir6.1. Nat Med 8, 466–472 (2002). https://doi.org/10.1038/nm0502-466
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0502-466
This article is cited by
-
Somatostatin-evoked Aβ catabolism in the brain: Mechanistic involvement of α-endosulfine-KATP channel pathway
Molecular Psychiatry (2022)
-
Propolis Promotes Memantine-Dependent Rescue of Cognitive Deficits in APP-KI Mice
Molecular Neurobiology (2022)
-
Cardiac dysfunction in spontaneously hypertensive old rats is associated with a significant decrease of SUR2 expression
Molecular and Cellular Biochemistry (2021)
-
Vascular KATP channels protect from cardiac dysfunction and preserve cardiac metabolism during endotoxemia
Journal of Molecular Medicine (2020)
-
Kir6.1 Heterozygous Mice Exhibit Aberrant Amygdala-Dependent Cued Fear Memory
Molecular Neurobiology (2020)